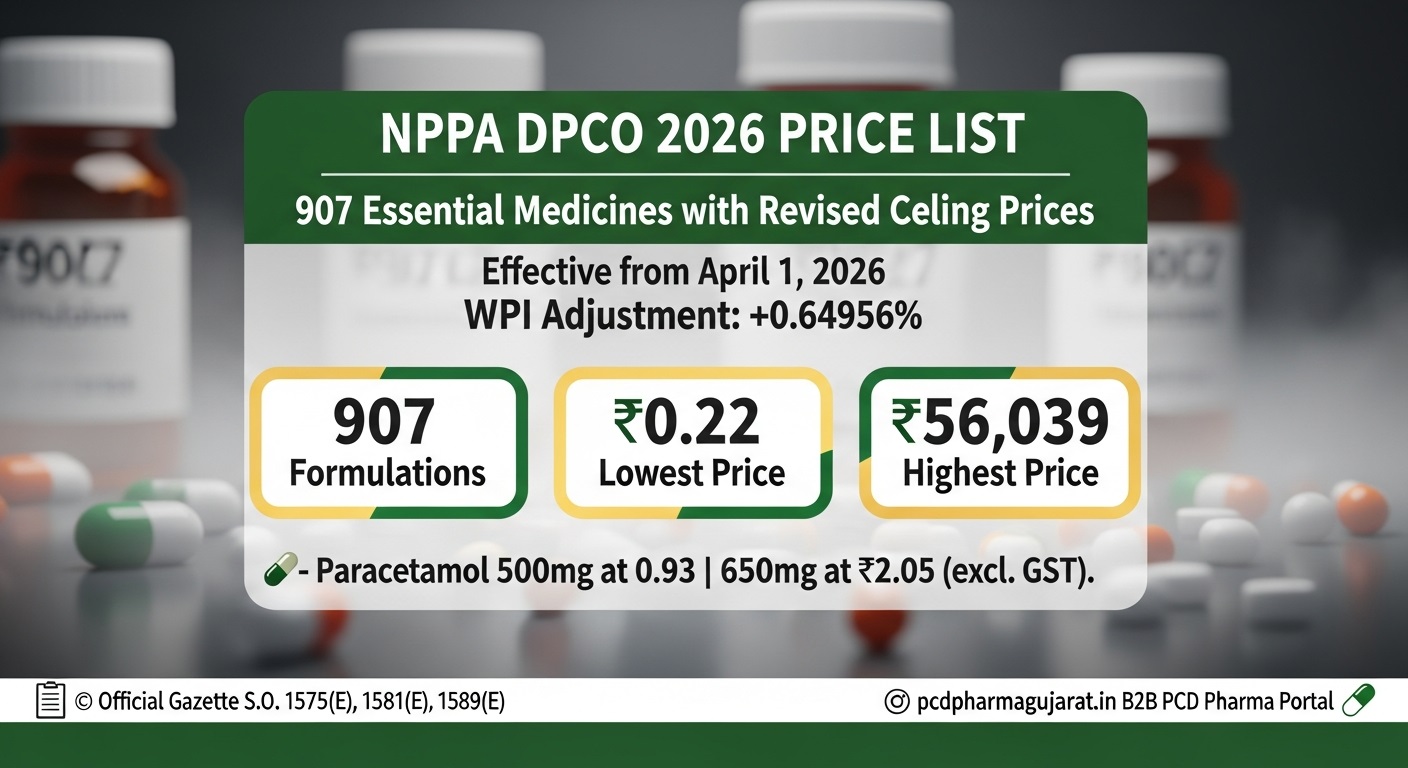
NPPA DPCO 2026 Price List | 907 Essential Medicines with Revised Ceiling Prices
Published: April 4, 2026 | Source: Gazette of India | Prepared by pcdpharmagujarat.in | NPPA DPCO 2026 Price List
Official NPPA DPCO 2026 Price List
Revised Ceiling Prices of Scheduled Formulations under Drug Price Control Order, 2013
Notifications: S.O. 1575(E), 1581(E), 1589(E) | Dated: March 25, 2026
Effective From: April 1, 2026WPI Adjustment: +0.64956%
Total Medicines: 907
• MRP must not exceed Ceiling Price + applicable GST
• Different pack sizes require separate NPPA approval
• Price list must be displayed at retail counters as per DPCO 2013
• Quarterly production & sales returns mandatory via IPDMS
Understanding the Latest NPPA Drug Price Revision 2026
The Government of India, through the National Pharmaceutical Pricing Authority (NPPA), has officially released the updated drug price list under the Drug Price Control Order (DPCO) 2013. This notification brings revised ceiling rates for 907 essential medicines, applicable nationwide from April 1, 2026.
The price revision incorporates an annual adjustment based on the Wholesale Price Index (WPI) of 0.64956% (2025 over 2024). This mechanism allows manufacturers to adjust for inflation while ensuring that critical drugs remain within affordable limits for the common citizen.
• First major revision of 2026 covering 767+ additional formulations
• Includes IV fluids, vaccines, anti-cancer drugs, and anti-retrovirals
• Dual-chamber bag formulations for Meropenem now under price control
• Paracetamol ceiling prices retained at ₹0.93 (500mg) and ₹2.05 (650mg)
What is DPCO and Why Does It Matter?
The Drugs (Prices Control) Order, 2013 (DPCO) is the principal legislation that empowers the Indian government to regulate the prices of essential medicines. Under this order, the NPPA fixes a ceiling price for drugs listed in the National List of Essential Medicines (NLEM).
For PCD pharma franchise businesses and stockists, staying updated with the latest DPCO list is crucial for:
- Compliance: Selling above the notified ceiling price attracts penalties and interest on overcharged amounts.
- Business Planning: Knowing price ceilings helps in procurement, pricing strategy, and margin calculation.
- Market Credibility: Distributors and retailers prefer dealing with franchise partners who maintain DPCO-compliant pricing.
Complete List of Scheduled Formulations (907 Medicines)
The table above contains the full, verified list of all scheduled formulations under the latest NPPA notification. This includes:
⚖️ Regulatory Framework: How NPPA Determines Ceiling Prices
The NPPA follows a scientific pricing methodology under DPCO 2013. The ceiling price for a scheduled formulation is calculated as the simple average of the prices of all brands having a market share of 1% or more, plus a 16% margin for retailers and distributors.
For the April 2026 revision, the authority applied the annual WPI increase of 0.64956% to the existing ceiling prices. This adjustment is permitted under paragraph 16(2) of DPCO 2013, which allows manufacturers to revise MRP annually based on WPI changes.
Key Examples from the 2026 Price List
| Medicine | Strength | New Ceiling Price (₹) |
|---|---|---|
| Paracetamol | 500mg Tablet | 0.93 |
| Paracetamol | 650mg Tablet | 2.05 |
| Amoxicillin | 500mg Capsule | 7.54 |
| Atorvastatin | 10mg Tablet | 5.09 |
| Metformin | 500mg Tablet | 2.07 |
| Insulin (Soluble) | 40 IU/ml Injection | 17.40 |
How to Verify Medicine Prices Using This List
For patients, pharmacists, and PCD franchise partners, verifying medicine prices is straightforward: Download List from links
- Add applicable GST (usually 5% or 12% depending on the drug category).
- Compare with MRP on the medicine strip — if higher, it may be a DPCO violation.
For official confirmation, you can visit the NPPA official website and download the complete gazette notification.
Download – Gazzet Copy:
https://nppa.gov.in/uploads/tender/0d883ca9b600c8a13cec452376176fc4.pdf
https://nppa.gov.in/uploads/tender/1a39a4415205950911f3893002626265.pdf
https://nppa.gov.in/uploads/tender/0fd8be342c35d5a46a13b281cadc5e4f.pdf
Implications for PCD Pharma Franchise Businesses
For our partners at pcdpharmagujarat.in, this price revision has several implications:
- Margin Recalculation: With new ceiling prices, franchise partners may need to adjust their retail margins while staying compliant.
- Inventory Management: Existing stock with older MRP must be repriced or returned to manufacturers.
- Documentation: Manufacturers must issue Form V price lists; distributors must display them prominently.
- Opportunity: Lower price ceilings on essential drugs can increase volume demand, benefiting franchise networks.
Important Deadlines for Manufacturers
- Price Revision: Effective immediately from April 1, 2026.
- Form V Filing: Price list must be submitted to NPPA via IPDMS portal from the date of notification.
- Overcharging Penalties: Any overcharged amount must be deposited with interest from the date of overcharging.
- Production Discontinuation: Manufacturers must notify NPPA at least six months in advance.
Conclusion of Blog on “NPPA DPCO 2026 Price List”
The NPPA’s DPCO 2026 price revision reflects the government’s ongoing commitment to making essential healthcare affordable. For the pharmaceutical industry, particularly PCD franchise networks like pcdpharmagujarat.in, staying compliant with these price ceilings is not just a legal obligation but also a mark of ethical business practice.
We recommend all our partners and readers to bookmark this page for future reference, as the table above contains the most current and complete list of scheduled formulations with their revised ceiling prices. Use the search functionality to quickly find any medicine, and always verify MRPs against these government-notified rates.
FOR PCD PHARMA FRANCHISE CLICK HERE & FILL FORM TE GET PCD PHARMA PRICE LIST.